Moderna’s May 8 stock jump appears to have been driven by a mix of real vaccine-pipeline news and speculative infectious-disease enthusiasm: the more meaningful catalyst was peer-reviewed Phase 3 mRNA flu vaccine data, while the hantavirus angle looks much more hype-driven and early-stage.
-
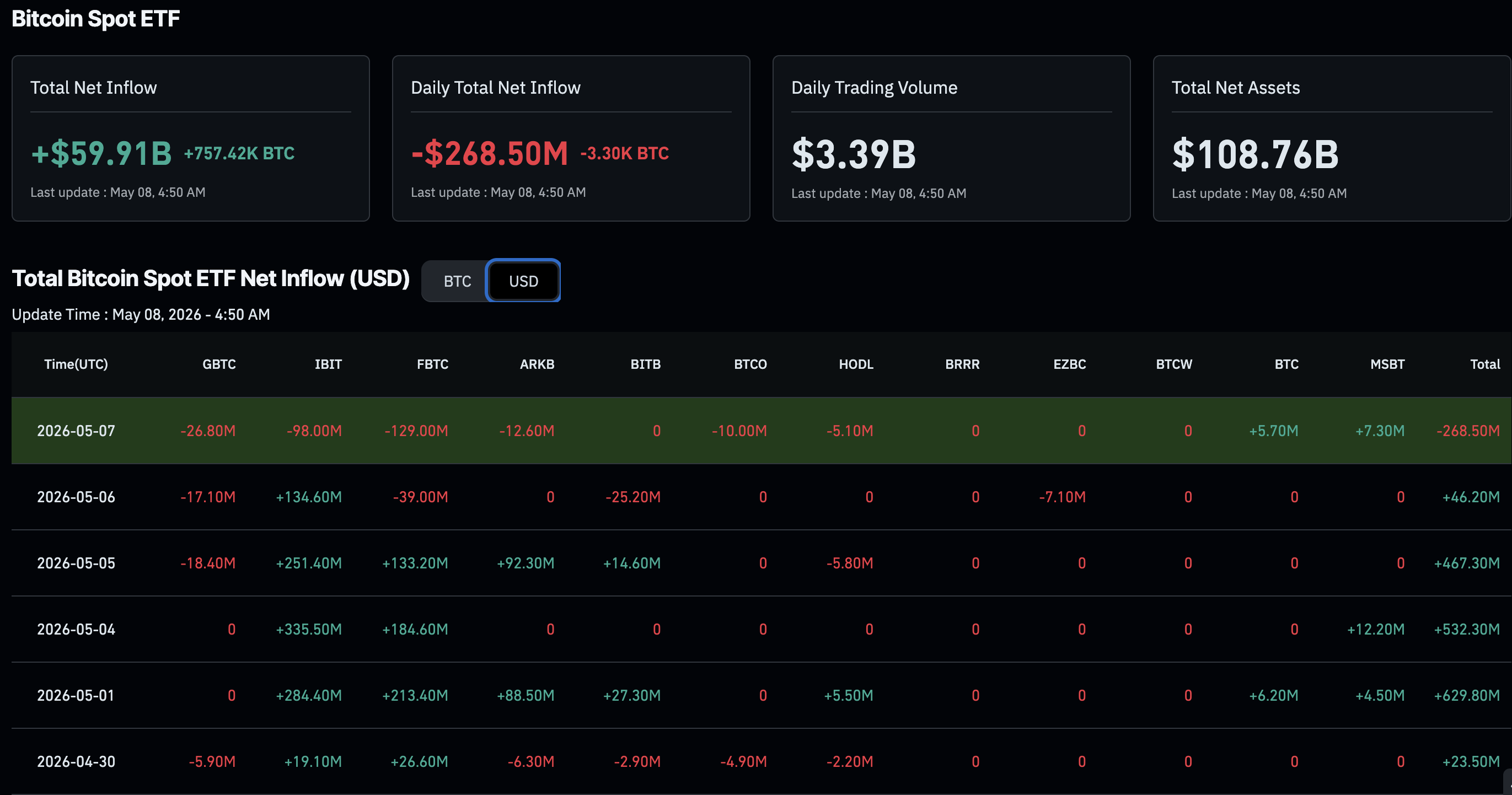
Moderna shares were reported up roughly 10%–16% on May 8, with coverage attributing the move to renewed focus on its mRNA pipeline, including hantavirus-vaccine reports and recently published flu-vaccine data [
9][
12].
-
The stronger fundamental news is the flu vaccine: Moderna announced that Phase 3 results for its investigational seasonal flu shot mRNA-1010 were published in the New England Journal of Medicine on May 6, 2026 [
6]. The study involved adults aged 50 and older, and a report on the trial said it enrolled more than 40,000 participants and showed mRNA-1010 was 26.6% more effective than a standard flu vaccine comparator [
10].
-
That flu data is commercially and medically significant because seasonal flu is a recurring, large vaccine market, and positive Phase 3 efficacy data can support regulatory approval and revenue diversification beyond COVID vaccines .